Aspergillus sp. in dwellings and
health implications of indoor fungi
Prepared for the Aspergillus website by:
Elena
Piecková,
Institute of Preventive and Clinical Medicine,
Limbová 14,
SK-833 01 Bratislava,
Slovakia. Email:pieckova@upkm.sk
According to his/her life style, a person can commonly spend 75
- 90 % of their time in an indoor environment. That is one reason
why the effects of different indoor factors on human health are
studied very carefully. Indoor bacteria or fungi can cause
allergic, infectious, toxic or inflammatory diseases i.
e. building-related illnesses. A complex of health troubles, bad
feelings and general discomfort due to a stay in certain buildings
is known as sick building syndrome (Kroeling, 1998).
Problems connected with the presence of microscopic fungi in
the working environment and their effects on the health status of
employees have been studied in many papers. It has been found that
the health of people working with moldy materials can be seriously
affected. The air of working environments may contain as much as
109 viable germs or particles (not only spores, also
hyphal fragments etc.) of microscopic filamentous fungi and yeasts
per m3 and a large amount of various
mycotoxins. Regarding the character of manufactured substrates,
irritation of eye, nose and mouth mucous membranes were found, as
well as serious acute and chronic damage of respiratory organs,
i.e. bronchitis, allergic alveolitis "farmer´s lung",
lung mycotoxicoses and similar disorders. Some mycotoxins can also
possess carcinogenic properties affecting lung tissue (Dutkiewicz,
1997, Dutkiewicz et al., 1994, Jesenská, 1993,
Sorenson et al., 1991).
While the work with moldy materials is
mostly sporadic and the possible adverse effects of the
microscopic fungi and their toxins can be foreseen and thus the
exposed persons can be protected using respiratory filters,
families including infants living in homes with moldy walls are
exposed to these noxae for a long periods of time. The amount of
fungal colony forming units (cfu)/m3 can vary from a
standard of 500 - 1000 to 6000 - 450000 cfu/m3 in
"moldy" houses (Nevalainen et al., 1991). Spores are
liberated into the air in places with higher
turbulence e.g. during home maintenance, cleaning,
dusting, vacuum cleaning, vegetable peeling, when door is opened,
when pets enter or leave etc. These activities can cause an
increase in air spore amount up to 3000-fold (Lehtonen and
Reponen, 1993). Many species of micromycetes (according to Hunter
et al. (1988) up to 56) have been isolated from
miscellaneous objects and walls in residences.
Allergies of inhabitants have been studied very extensively in
connection with moldy dwellings. Cytoplasmic glycoproteins of
fungal spores are important aeroallergens in genetically
pre-determined humans (Linas et al., 1998). In dwellings
occupied by children suffering from asthma, significantly greater
spore counts of Cladosporium sp. and Penicillium
sp. were found (Li et al., 1995).In some case studies,
explicit relations between the presence of the fungus in dwellings
and allergic reactions of their occupants were found. After
cleaning the contaminated sites, or when the occupants moved into
a new home, the symptoms disappeared (Senkpiel et al.,
1996).
It was found that allergic alveolitis (hypersensitivity
pneumonia) was caused by Penicillium expansum (Park et
al., 1994) and Rhodotorula rubra (Siersted and
Gravesen, 1993). Kanny et al. (1996) found the cause of an
eczema and respiratory disorder in a 25-years-old female student
was her hypersensitivity against the antigens of micromycetes
growing on walls of her home, i.e. Fusarium sp., Cladosporium sp., Pullularia sp., Rhizopus
sp. and Penicillium sp. An explicit relation between summer
hypersensitivity pneumonia and the presence of Trichosporon
sp. in homes of the patients was found in Japan (Ando et
al., 1991, Lehtonen and Reponen, 1993, Rylander, 1997, Sumi
et al., 1994, Summerbell et al., 1992). Kauffman
et al. (1995) found that atopic humans suffered from acute
symptoms of fungal asthma when they inspired a great number of
spores. An adverse situation appears when mucous membrane of the
epithelium of respiratory tract of sensitive person is
systematically colonized with fungi whose cells possess the
ability to penetrate into the deeper parts of lung tissue and
produce protease in a depository niche. These authors also found
that sensitization with fungal antigens is dependent on the
person´s age, as most humans with positive skin tests to the
antigens of micromycetes are persons in younger age categories. An
increasing mortality among asthmatic patients was observed during
the period when a statistically significant higher number of
fungal spores was found in the air (Targonski et al.,
1995). According to Garrett et al. (1998), the high
incidence of Penicillium sp. in dwellings was a significant
risk factor for asthma, while spores of Aspergilli potentiated
atopic reactions of inhabitants. Based on long-term
epidemiological studies in USA, Australia, New Zealand and the
European Nordic Countries, the statistic significance of higher
outbreak of respiratory allergies (asthma, allergic rhinitis,
hypersensitivity pneumonia) in children from damp and wet
dwellings with a high concentration of fungal, namely
Alternaria sp. spores in the air, was apparent (Huang and
Kimbrough, 1997, Page and Trout, 1998, Peat et al.,
1998).
There is a close relationship between the presence of spores of
microscopic fungi in the indoor air of dwellings and allergic
symptoms in some patients. Nevertheless, most of the patients
suffer from "multiallergy", with allergic reaction also
against other environmental components. The relationship between
the fungal antigens and the allergic patients is considerably
influenced by the personality of the patient. This probably causes
in some groups of atopic humans an apparently clear relation
between the spore count in the air and the patient´s reaction, but
this could not be proved (Dill and Niggemann, 1996, Jarvis et
al., 1996). For the mycologists, there is
a great challenge in preparing a high quality antigen test
sets to prove patient´s allergy.
Currently, a general approach to the study of the mechanism of
fungal effects on human beings is becoming more urgent. Such an
approach includes the immunosuppressive influence of beta-glucans
from fungal cell wall as well as toxic and irritative effects of
secondary metabolites - mycotoxins and/or volatile organic
compounds (Larsen et al., 1998). Low molecular organic
compounds, namely alcohols, aldehydes, ketones, aromatic
compounds, amines, terpenes, chlorinated hydrocarbons and
sulphuric compounds, cause typical "moldy" odor but also an
inflammation of airways of sensitive people. Aspergillus
sp., A. versicolor, Cladosporium sp. and Penicillium
sp. are strong producers of these compounds. Such effects are
associated with invisible moulds growing under wallpaper, carpets
or mattresses (Korpi et al., 1997). In some well-known
toxinogenic strains, a direct correlation was found between
the mycotoxin production and production of volatile products,
e.g. ketones (Pasanen et al., 1996).
Some authors have revealed that in occupants of
"moldy" dwellings there is a higher incidence of
bronchitis, sore throat, concentration difficulties, back-aches,
irritation of eyes and mouth cavity, feeling of weekness
etc. (Pirhonen et al., 1996, Summerbell et al.,
1992). Ill health symptoms were also associated with increased
amounts of Epicoccum sp., Aureobasidium sp. and
yeasts in dwellings (Su et al., 1992). Chronic intoxication
is also suffered by the occupants of a house where the air
conditioning system and wet ceiling were contaminated with
Stachybotrys chartarum, and in indoor air there were found
spores of that species (Bjurman and Kristensson, 1992). A study
was carried out on an unusual incidence of acute lung
hemosideroses in infants aged six weeks to six months in
Cleveland, Ohio, USA. One child died, and the only common sign was
that all those families lived in dwellings contaminated with
S. chartarum. Among others, Memnoniella echinata
strains were also isolated from the environment, and it was found
that their metabolites were highly cytotoxic. It was also proved
that the M. echinata strains produce trichothecenous
mycotoxins trichodermol and trichodermin. Jarvis et
al. (1996) reported that this species can sometimes grow
together with S. chartarum. Massive incidence of spores of
S. chartarum was also found in the indoor work environment
which had been flooded accidentally. The staff (53 persons)
suffered from symptoms of toxic damage of the lower part of lung
tissue, skin and eyes irritations and chronic
weakness. Statistically significant damage to the immune system,
especially of T-lymphocytes was found too (Johanning et
al., 1996). S. chartarum strains produce toxic
trichothecenes, e.g. satratoxins, verrucarins, roridins and
others. The toxic metabolites stachybotryotoxins-trichothecenes
are concentrated in the cells of the fungus, they are produced in
phialides, conidia, conidiophores but also diffuse into growth
medium (Pasanen et al., 1993).
An occupant of a "moldy" dwelling is exposed in that
environment not only to microscopic fungi but also to a number of
volatile compounds released from furniture, carpets, various
paints and other materials. The mechanism of the potential
pathological effects of microscopic fungi and their toxins on lung
tissue in healthy humans has not yet been made sufficiently
clear.
For estimating the micromycete effects in the indoor home
environments it is necessary to consider the results of the
experiments conducted in vitro or on laboratory
animals. Thus, it was found that 47 % of the examined strains
isolated from the dwellings were cytotoxic against human embryonal
diploid fibroblasts from lung tissue (Smith et al.,
1992). The pure known toxic metabolites of microscopic fungi, e.g.
trichothecenes and sterigmatocystin, and extracts from some
strains isolated from house walls cause cilia of tracheal mucous
membranes to stop moving in organ cultures from one-day-old
chickens and affect the self-cleaning ability of these membranes
(Jesenská and Bernát, 1994, Piecková and
Jesenská, 1995, 1996, 1997a, b, 1998). Intranasal
application of 1x106 spores of the satratoxin-producing
S. chartarum strain caused inflammatory reactions in the
lung tissue of experimental mice together with exudative processes
in alveoli and bronchi. These disorders could be classified
as lung mycotoxicosis (Nikulin et al., 1996).
Characteristics and properties of Aspergilli in the indoor
environment
Regarding the health conditions of the occupants whose
dwellings have moldy walls, it is important to pay special
attention to the species of Aspergillus clavatus,
A. fumigatus, species from the group of A. niger,
and A. versicolor, rarely to other fungi of this
genus.
Indoor air in dwellings. In the USA, the
Aspergillus sp. fungus was found in 9 - 29 % and in Scotland
in 74 % of the examined homes. In Ontario, Canada, their
concentration in the air of dwellings was 22 cfu/m3.
In Slovakia, Aspergillus sp. strains were isolated from 9 %,
A. flavus from 30 % ( 5 % of strains were able to produce
alflatoxins B1, B2, G1 and G2
in vitro), Aspergilli from the group of A. glaucus
from 2 %, A. ochraceus from 12 %, Aspergilli from the group
of A. restrictus from 2 % and A. ustus from 5 % of
examined "moldy" dwellings. Microscopic pictures of
scrapings from walls and wooden window frames of some houses are
presented (Fig. 1-4) (Piecková et al., 1999).
|
|
|
|
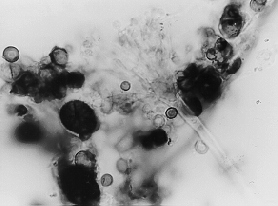
Fig 1. Scraping from a wall in a
nursery - a fragment of a fruiting body of
A. versicolor type (magn. 1000x).
|
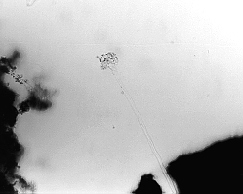
Fig 2. Scraping from a wall in a
fitness room - a fruiting body of Aspergillus
sp. type (magn. 200x).
|
|
|
|
|
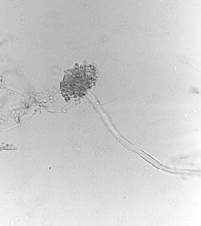
Fig 3. Home dust - a fruiting body of
A. versicolor (magn. 400x).
|
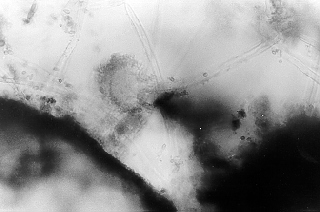
Fig 4. Scraping from the window frame
in a fitness room - a fruiting body of Aspergillus sp. type (magn. 400x).
|
During the evaluation of dwellings in Belgium, the following
Aspergilli were isolated: A. fumigatus from kitchens and
bathrooms, A. versicolor and A. repens from
mattresses and carpets, A. versicolor and
A. fumigatus from cellars, A. fumigatus,
A. niger and A. flavus from flower pot soil, A. versicolor andA. fumigatus from various pad
materials (Summerbell et al., 1992). In Denmark, Aspergillus sp. strains were isolated from 56 % of evaluated
moldy indoor samples (Gravesen et al., 1999).
Damp materials with an aw value ranging from 0.90 to
0.95 are usually colonized by strains of A. fumigatus,
materials with aw values ranging from 0.90 to 0.85 by
strains of A. versicolor, materials with aw
values of 0.85 and a little less, by strains of A.
versicolor, Eurotium sp. (Samson et al., 1994).
In general, it is thought that the amount of Aspergillus
sp. germs is higher in indoor air than outdoors at any
given time. In the home, the amount of spores in the air is
markedly increased when the cleaning is carried out mechanically,
for example, when carpets are vacuum cleaned.
A. clavatus is often associated with allergic
alveolitis (hypersensitivity pneumonia) in workers of malthouses
(Reynolds, 1991) . >Minimum awvalue for its growth is
0.85.
A. fumigatus is the most important and well known
potential pathogen for humans with affected immunity.
Allergic diseases. As much as 10.8 % of asthma patients
had positive reactions to the extract from A. fumigatus
strains. Nevertheless, A. fumigatus possesses similar
biochemical structure and antigenic properties to Penicillium
glabrum, P. verrucosum var. verrucosum and
A. versicolor. A. fumigatus spores have the ability
to be bound on lung epithelium in asthma patients, causing
complications in the health status of the patients (Bromley and
Donaldson, 1996). In that case, the course of the patients
disease assumes a very destructive character (Kauffman et
al., 1995).
Aspergillus Sp. were found in indoor air of
dwellings, e.g. in South California in 2.9 % and in the
Netherlands in 4.5 % of examined dwellings. In wallpapers of a
hotel in Singapore, its incidence was as high as 18 - 88 %. In an air
conditioning system in Saudi Arabia, it formed 11 % of all fungal
isolates and its concentration was 15 700 cfu/g of dust.
When examining materials from the indoor work environment, it
was found that about 70 % of A. fumigatus spores were able -
due to their size - to penetrate into the trachea and primary
bronchi and less than 1 % into alveoli (Millner et al.,
1980).
The best known toxic metabolites of A. fumigatus are
mainly fumigaclavin A, B, C, and D, spinulosin and tremorgenous
toxins, e.g. verruculogen. Some rarely isolated strains produce
kojic acid, sterigmatocystin and various unknown toxins affecting
Artemia salina and Bacillus megatherium NRRL 1366,
chicken embryos and other organisms. From 106 spores of
the A. fumigatus SRRC 2006 strain, an amount of 9.89 ng of
fumigaclavin A was isolated. In five out of eight
A. fumigatus strains isolated from the indoor work
environment in a sawmill, fumigaclavin C and verruculogen were
found (Land et al., 1987). The investigation was carried
out in connection with the assumed lung mycosis in workers. In the
filtrate from mycelium and medium the presence of the hemolytic
toxin was demonstrated, containing large protein amount and traces
of saccharides (Yokota et al., 1977).
Minimum temperature for mycelium growth is 10 -
12oC, optimum 37 - 43oC, minimum relative
air humidity 85 %, optimum 98 % and for conidia formation minimum
90 %, optimum 98 - 99 %. Minimum aw value for A. fumigatus growth is 0.94 and for sporulation 0.95. No
spore germination was observed at 4 - 8oC, optimum
germination temperature is 25oC and optimum aw
value is 0.94. Optimum pH value of the environment for
A. fumigatus ranges from 3.0-8.0.
A. niger. Aspergilli, characteristic with their
striking black pigmentation of the colonies, are classified within
a large A.niger group. In most papers describing the
isolation of such strains, they are presented simply as
A. niger. However, in that case it is difficult to
differentiate which species were involved in the activity
described below in the indoor environment and in the pathologic
process.
Indoor air in the dwellings. A. niger isolates
were described in homes in London and Central Scotland, in
Plzeň (Czech Republic)
(0.71 - 1.88 % of all isolates) and in Egypt (15 % of all
isolates). They were found in Saudi Arabia in the dust from an air
conditioning system (10 600 cfu/g and 7.93 % of all isolates) and
in dust from dwellings (8 600 cfu and 4 % from all isolates), in
homes in California (in 19 % of homes, on the average 2.9
cfu/m3, maximum 59 cfu/m3), in Canada (in 6
% of homes), in Ontario, Canada, (on the average 7
cfu/m3). Results presented in another paper describe
the presence of A. niger in the dust from Canadian homes
in 50 % of all examined samples in the amount of 0.7x104
cfu/g. In the Netherlands, it was found in 4.5 % of
homes, in Slovakia, in 5 % of "moldy" dwellings.
The fungus is acidophilic, the mimimum pH value of 1.5 is
suitable for mycelium growth, optimum pH is 4.4 - 7.5, maximum
9.8. Minimum temperature 6 - 8°C, optimum
35 - 37°C, maximum 45 - 47°C. Minimum air
humidity is 88 - 89 %, optimum 96 - 98 %, and minimum aw
value is 0.85. Minimum humidity for conidia formation is
92 - 95 %, optimum 96 - 98 %. Conidia germinate only in temperatures
exceeding 10°C.
A. niger strains are known to be producers of
many enzymes and other metabolites . To the well known toxic
metabolites belong the malformins A1, A2,
B1, B2, C and oxalic acid. In the spores of
the A. niger strain SRRC 2005, aurasporon C was
detected. However, the papers describing the incidence of toxic
strains are full of contradictions that may be due to the
imprecise identification of the isolates, as mentioned above.
A. versicolor has an antigen structure similar to
that of P. glabrum. After an inhalation of A.
versicolor spores, laboratory rats showed granulomatous
lesions in lung tissue, localized mainly near to the bronchi,
after one month exposure (Sumi et al., 1994).
Indoor air in the dwellings. A. versicolor was
found in indoor air of homes in London, in Central Scotland, and
in the Netherlands (in 9 of 11 examined homes, in 32 % of all
examined homes, and in 50 % of examined schools). It formed 16 -
27% of isolates in homes in Torino, Italy, and 11.5 % of isolates
from moldy wallpapers. A. versicolor was found also in dust
in dwellings in Saudi Arabia (15 000 cfu/g and 7.66 % of all
isolates). In Slovakia, it was isolated from 33 % of examined
"moldy" dwellings. This fungus is placed among the primary wall
colonizers at 25°C in dwellings and was also found in dust from
mattresses.
A.versicolor spores germinate in 12 - 20 days in
dependence on the temperature and the pH values of substrates and
at the aw values ranging from 0.75 to 0.81. The aw
values reported for this species are within the range
0.78 - 0.98, minimum growth temperature 6 - 9°C,
and optimum temperature is 25 - 27°C. An
A. versicolor isolate from moldy house wall was able to
survive incubation onto six different types of plaster at 25°C in
an atmosphere saturated with moisture for three months
(Piecková and Jesenská, 2000).
Most of the relevant papers deal with the production of toxic
and carcinogenic strigmatocystin by A. versicolor. The
amount of toxinogenic strains is rather high, about 74% out
of strains tested produced this toxin under various laboratory
conditions. Strains of A. versicolor isolated from
damp houses in USA, with occupants suffering from pulmonary
disease, were able to produce this mycotoxin in vitro, too
(Hodgson et al., 1998). A. versicolor isolates from
"moldy" dwellings cultivated on plasterboard at 25°C in an
atmosphere saturated with moisture for 46 - 77 days produced
chloroform-extractable endo- and exometabolites able to stop the
movement of cilia of chicken tracheal epithelium, similar to the
effect of sterigmatocystin (Piecková et al.,
1999).
Monitoring of exposure to the indoor fungi is rather
complicated due to lack of standard and exact practical methods to
evaluate how indoor microclimate (temperature, relative humidity,
dust, ventilation, constructing materials and furnishings),
outdoor ambient, and microscopic fungi affect each other.
To determine a causal relationship between mycotoxins in the
indoor environment and particular human health disorders it is
necessary to quantify mycotoxin concentration in damp and
"healthy" buildings, to estimate minimal concentration of
mycotoxin able to cause clinical symptoms in comparison with in vitro models, to choose optimal animal or other biological
models for studying of mycotoxin pathogenicity and pathophysiology
and to characterize short- and long-term health damages (not only
biomarkers) in people under such an influence (Garrett et
al., 1998).
Fungal contamination of the indoor environment of buildings has
to be solved at the level of building, maintenance, and style of
life and health of the occupants. This contamination is an
indicator of unsuitable architecture, building construction and/or
using of building. To reduce the concentration of microscopic
fungi in the indoor environment, reduction of dampness, effective
ventilation and right regime of temperature and hygiene is
recommended. In particular cases, it is useful to consult
industrial hygiene experts.
REFERENCES
Ando M, Arina K, Yoneda R, Tamura M. Japanese summer-type
hypersensitivity pneumonitis - geographic distribution, home
environment and clinical characteristics of 621 cases. American
Revue of Respiratory Diseases 1991;144:765-9.
Bjurman J, Kristensson J. Volatile production by Aspergillus
versicolor as a possible cause of odor in houses affected by
fungi. Mycopathologia 1992;118:173-8.
Bromley IMJ, Donaldson K. Binding of Aspergillus
fumigatus spores to lung epithelial cells and basement
membrane proteins: relevance to the asthmatic lung. Thorax
1996;51:1203-9.
Dill I, Niggemann B. Domestic fungal viable propagules and
sensitization in children with IgE mediated allergic
diseases. Pediatric Allergy and Immunology 1996;7:151-5.
Dutkiewicz J. Bacteria and fungi in organic dust as potential
health hazard. Annals of Agricultural and Environmental Medicine
1997;4:11-6.
Dutkiewicz J, Pomorski ZJH, Sirkowska J, Krysinska-Traczyk E,
Skórska C, Prazmo Z, Cholewa G, Wójtowicz H. Airborne
microorganisms and endotoxin in animal houses. Grana
1994;33:85-90.
Garrett MH, Rayment PR, Hooper MA, Abramson MJ, Hooper
BM. Indoor airborne fungal spores, house dampness and associations
with environmental factors and respiratory health in
children. Clinical and Experimental Allergology
1998;28:459-67.
Gravesen S, Nielsen PA, Iversen R, Nielsen KF. Microfungal
contamination of damp buildings - examples of risk constructions
and risk materials. Environmental Health Perspectives
1999;107:505-8.
Hodgson MJ, Morey P, Leung W-Y, Morrow L, Miller D, Jarvis BB,
Robbins H, Halsey JF, Storey E. Biulding-associated pulmonary
disease from exposure to Stachybotrys chartarum and Aspergillus versicolor. Journal of Occupational and
Environmental Medicine 1998;40:241-9.
Huang S-W, Kimbrough JW. Mold allergy is a risk factor for
persistent cold-like symptoms in children. Clinical Pediatrics
1997;36:695-9.
Hunter CA, Grant C, Flanningan B, Bravery AF. Mould in
building: the air spora of domestic dwellings. International
Biodeterioration 1988;24:81-101.
Jarvis BB, Zhou Z, Jiang J, Wang S, Sorenson WG, Hintikka EL,
Nikulin M, Parikka P, Etzel RA, Dearborn DG. Toxigenic molds in
water-damaged buildings: dechlorogriseofulvins from Memnoniella
echinata. Journal of Natural Products 1996; 59:553-4.
Jesenská Z. Micromycetes in Foodstuffs and
Feedstuffs. 1st ed. Amsterdam: Elsevier; 1993.
Jesenská Z, Bernát D. Effect of mycotoxins on
in vitro movement of tracheal cilia from one-day-old
chicks. Folia Microbiologica 1994;39: 155-8.
Johanning E, Biagini R, Hull D, Morey P, Jarvis B, Landsbergis
P. Health and immunology study following exposure to toxigenic
fungi (Stachybotrys chartarum) in water-damaged office
environment. International Archives of Occupational and
Environmental Health 1996;68:207-18.
Kanny G, Becker S, de Hauteclocque C, Moneret-Vautrin
DA. Airborne eczema due to mould allergy. Contact Dermatitis
1996;35:378.
Kauffman HF, Tomee JFC, van der Werf TS, de Monchy JGR, Koeter
GK. Review of fungus-induced asthmatic reaction. American Journal
of Respiratory and Critical Care Medicine 1995;151:2109-16.
Korpi A, Pasanen A-L, Pasanen P, Kalliokoski P. Microbial
growth and metabolism in house dust. International
Biodeterioration and Biodegradation 1997;40.19-27.
Kroeling P. Sick building syndrom. Allergy 1998;21:180-91.
Land CJ, Hult K, Fuchs R, Hagelberg S, Lundström H. Tremorgenic
mycotoxins from Aspergillus fumigatus as a possible
occupational health problem in sawmills. Applied and Environmental
Microbiology 1987;53:787-90.
Larsen FO, Clementsen P, Hansen M, Maltbaek N, Ostenfeldtlarsen
T, Nielsen KF, Gravesen S, Skov PS, Norn S. Volatile organic
compounds from the indoor mould Trichoderma viride cause
histamine release from human bronchoalveolar cells. Inflammatory
Research 1998;47:S5-6.
Lehtonen M, Reponen T. Every activities and variation of fungal
spore concentrations in indoor air. International Biodeterioration
and Biodegradation 1993; 31:25-40.
Li C-S, Hsu L-Y, Chou C-C, Hsieh K-H. Fungus allergens inside
and outside the residences of atopic and control
children. Archives of Environmental Medicine 1995; 50:38-43.
Linas MD, Morassin B, Recco P. Current data on
Alternaria. Revue of French Allergology 1998;38.349-55.
Millner PD, Bassett DT, Marsh PB. Dispersal of Aspergillus
fumigatus from sewage sludge compost piles subjected to
mechanical agitation in open air. Applied and Environmental
Microbiology 1980;39:1000-9.
Nevalainen A, Pasanen AL, Niininen M, Reponen T, Kalliokoski P,
Jantunen MJ. The indoor air quality in Finnish homes with mold
problems. Environment International 1991;17:299-302.
Nikulin M, Reijula K, Javis BB, Hintikka E-L. Experimental lung
mycotoxicosis in mice induced by Stachybotrys atra.
International Journal of Experimental Pathology
1996;77:213-8.
Page E, Trout D. Mycotoxins and building related
illness. Journal of Occupational and Environmental Medicine
1998;40:761-3.
Park H-S, Jung K-S, Kim SO, Kim SJ. Hypersensitivity
pneumonitis induced by Penicillium expansum in a home
environment. Clinical and Experimental Allergology
1994;24:383-5.
Pasanen A-L, Lappalainen S, Pasanen P. Volatile organic
metabolites associated with some toxic fungi and their
mycotoxins. Analyst 1996;121:1949-53.
Pasanen A-L, Nikulin M, Toumainen M, Berg SY, Parikka P,
Hintikka, EL. Laboratory experiments on membrane filter sampling
of airborne mycotoxins produced by Stachybotrys atra
Corda. Atmospheric Environment 1993;27A:9-13.
Peat JK, Dickerson J, Li J. Effects of damp and mould in the
home on respiratory health: a review of the literature. Allergy
1998;53:120-8.
Piecková E, Jesenská Z. The effect of
chloroform-extractable secondary metabolites of filamentous fungi
on the movement of respiratory tract cilia of one-day-old chicks
in vitro. Folia Microbiologica 1995;40:123-7.
Piecková E, Jesenská Z Ciliostatic effect of
fungi on the respiratory tract ciliary movement of one-day-old
chickens in vitro. Folia Microbiologica
1996;41:517-20.
Piecková E, Jesenská Z. Ciliostatic activity in
day-old chicks indicates microscopic fungi toxicity in
vitro. Annals of Agricultural and Environmental Medicine
1997;4: 35-6.
Piecková E, Jesenská Z. The effect of chloroform
extracts of micromycete biomass on the movement of tracheal cilia
in one-day old chickens in vitro. Czech Mycology
1997;50:57-62.
Piecková E, Jesenská Z. Molds on house walls and
the effect of their chloroform-extractable metabolites on the
resipratory cilia movement of one-day-old chiks in
vitro. Folia Microbiologica 1998;43:672-8.
Piecková E, Jesenská Z. Investigation of mycotic
contamination of plasters in vitro. (In Slovak). Hygiena
2000;45:37-31.
Piecková E, Jesenská Z, Sternová
Z. Microscopic filamentous fungi (molds). In: Sternová Z,
editor. Injurants damaging health in dwellings. (In
Slovak). Bratislava: Zing Print; 1999. P. 7-53.
Pirhonen I, Nevalainen A, Husman T, Pekkanen J. Home dampness,
moulds and their influence on respiratory infections and symptoms
in adults in Finland. European Respiratory Journal
1996;9:2618-22.
Reynolds HY. Hypersensitivity pneumonitis. Correlation of
cellular and immunologic changes with clinical phases of
disease. Lung 1991;169:S129-30.
Rylander R. Airborne (1-3)-beta-D-glucan and airway disease in
a day-care center before and after renovation. Archives of
Environmental Health 1997;52:281-5.
Samson RA, Flannigan B, Flannigan ME, Verhoeff AP, Adan OCG,
Hoekstra ES. Health implications of fungi in indoor
environments. 1st ed. Amsterdam: Elsevier; 1994.
Senkpiel K, Kurowski V, Ohgke H. Investigation of fungal
contamination of indoor air in homes of selected patients with
asthma bronchiale. Zentralblatt fur Hygiene 1996;198:191-203.
Siersted HC, Gravesen S. Extrinsic allergic alveolitis after
exposure to the yeast Rhodotorula rubra. Allergy
1993;48:298-9.
Smith JE, Anderson JG, Lewis CW, Murad YM. Cytotoxic fungal
spores in the indoor atmosphere of the damp domestic
environment. FEMS Microbiological Letters
1992;100:337-43.
Sorenson WG, Simpson J, Dutkiewicz J. Yeasts and yeast-like
fungi in stored timber. International Biodeterioration
1991;27:373-82.
Su HJ, Rotnitzky A, Burge HA, Spengler JD. Examination of fungi
in domestic interiors by using factor analysis: correlations and
associations with home factors. Applied and Environmental
Microbiology 1992;58:181-6.
Sumi Y, Nagura H, Takeuchi M, Miyakawa M. Granulomatous lesions
in the lung induced by inhalation of mold spores. Virchow Archive
1994;424:661-8.
Summerbell RC, Staib F, Dales R, Nolard N, Kane J, Zwanenburg
H, Burnett R, Krajden S, Fung D, Leong D. Ecology of fungi in
human dwellings. Journal of Medical and Veterinary Mycology
1992;Suppl.1:279-85.
Targonski PV, Persky VW, Ramekrishman V. Effect of
environmental molds on risk of death from asthma during the pollen
season. Journal of Allergology and Clinical Immunology
1995;95:955-61.
Yokota K, Shimada H, Kamaguchim A, Sakaguchi O. Studies of the
toxin of Aspergillus fumigatus. VII. Purification and some
properties of hemolytic toxin (Asp-hemolysin) from culture
filtrate and mycelia. Microbiology and Immunology 1977;21:
11-22.



